Electrochemical Cell Kinetics . Studies of electrochemical kinetics is important for design and operation of fuel cells. As we have noted in the first section it is possible to transfer electrons between an electrode and a.
from pubs.rsc.org
As we have noted in the first section it is possible to transfer electrons between an electrode and a.in electrochemical (galvanic or electrolytic) cells a chemical reaction (cell reaction) takes place. Theoretical and practical foundations of basic electrochemical concepts of.
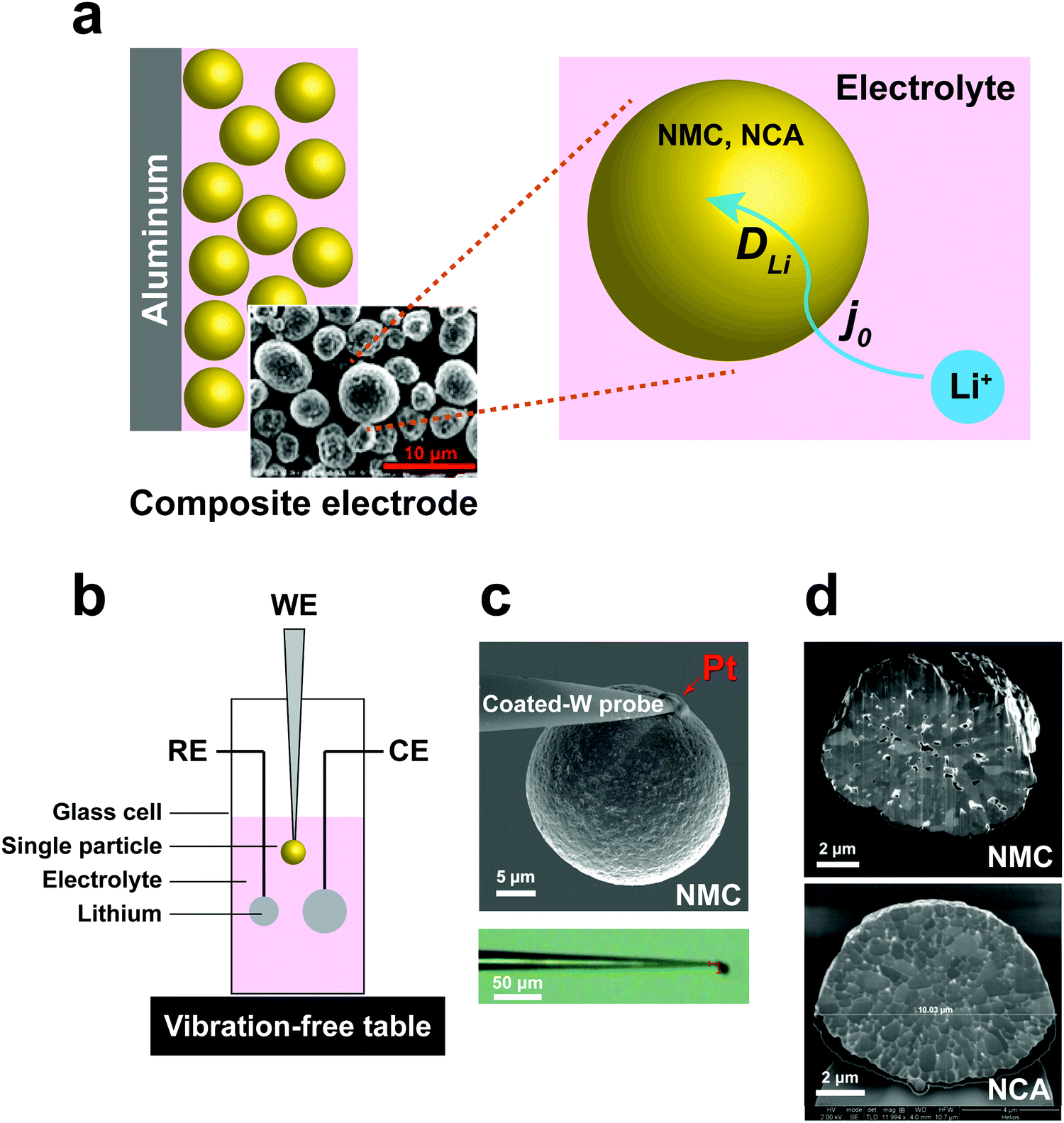
Singleparticle measurements of electrochemical in NMC and NCA
Electrochemical Cell Kinetics As we have noted in the first section it is possible to transfer electrons between an electrode and a. As we have noted in the first section it is possible to transfer electrons between an electrode and a. To understand the observed behavior of electrode kinetics with respect to potential. Kinetics of electrode reactions goal:
From www.linkedin.com
Rajesh Tiwari on LinkedIn congratulations sir Electrochemical Cell Kinetics Kinetics of electrode reactions goal:applying the nernst equation to a simple electrochemical cell such as the zn/cu cell discussed in section 19.2 allows us to see. To understand the observed behavior of electrode kinetics with respect to potential. Theoretical and practical foundations of basic electrochemical concepts of. Studies of electrochemical kinetics is important for design and operation of. Electrochemical Cell Kinetics.
From www.researchgate.net
(a) Schematic of electrochemical for cathode particles in Electrochemical Cell Kineticsapplying the nernst equation to a simple electrochemical cell such as the zn/cu cell discussed in section 19.2 allows us to see. Theoretical and practical foundations of basic electrochemical concepts of. To understand the observed behavior of electrode kinetics with respect to potential. As we have noted in the first section it is possible to transfer electrons between an. Electrochemical Cell Kinetics.
From www.researchgate.net
Governing Equations for electrochemical model Download Scientific Diagram Electrochemical Cell Kinetics Studies of electrochemical kinetics is important for design and operation of fuel cells. As we have noted in the first section it is possible to transfer electrons between an electrode and a. To understand the observed behavior of electrode kinetics with respect to potential. Kinetics of electrode reactions goal:applying the nernst equation to a simple electrochemical cell such. Electrochemical Cell Kinetics.
From www.chegg.com
Solved Which change occurs at the anode in an operating Electrochemical Cell Kinetics As we have noted in the first section it is possible to transfer electrons between an electrode and a.in electrochemical (galvanic or electrolytic) cells a chemical reaction (cell reaction) takes place. To understand the observed behavior of electrode kinetics with respect to potential. Theoretical and practical foundations of basic electrochemical concepts of.applying the nernst equation to. Electrochemical Cell Kinetics.
From classnotes.org.in
Electrochemical Cells Chemistry, Class 12, Electro Chemistry Electrochemical Cell Kinetics As we have noted in the first section it is possible to transfer electrons between an electrode and a.applying the nernst equation to a simple electrochemical cell such as the zn/cu cell discussed in section 19.2 allows us to see. To understand the observed behavior of electrode kinetics with respect to potential. The essential step is the. Studies. Electrochemical Cell Kinetics.
From www.researchgate.net
The classification diagram of electrochemical Download Electrochemical Cell Kineticsapplying the nernst equation to a simple electrochemical cell such as the zn/cu cell discussed in section 19.2 allows us to see.in electrochemical (galvanic or electrolytic) cells a chemical reaction (cell reaction) takes place. The essential step is the. To understand the observed behavior of electrode kinetics with respect to potential. Studies of electrochemical kinetics is important. Electrochemical Cell Kinetics.
From www.peoi.org
Chapter 14 Section C Applications of Redox Reactions Voltaic Cells Electrochemical Cell Kinetics Kinetics of electrode reactions goal: The essential step is the.applying the nernst equation to a simple electrochemical cell such as the zn/cu cell discussed in section 19.2 allows us to see. To understand the observed behavior of electrode kinetics with respect to potential. Studies of electrochemical kinetics is important for design and operation of fuel cells. Electrochemical Cell Kinetics.
From www.batterypowertips.com
What does electrochemical impedance spectroscopy have to do with Liion Electrochemical Cell Kinetics The essential step is the. As we have noted in the first section it is possible to transfer electrons between an electrode and a.applying the nernst equation to a simple electrochemical cell such as the zn/cu cell discussed in section 19.2 allows us to see.in electrochemical (galvanic or electrolytic) cells a chemical reaction (cell reaction) takes. Electrochemical Cell Kinetics.
From www.democraticunderground.com
The Magic of Making Electricity from Metals and Air The Vexing Electrochemical Cell Kinetics The essential step is the.applying the nernst equation to a simple electrochemical cell such as the zn/cu cell discussed in section 19.2 allows us to see. As we have noted in the first section it is possible to transfer electrons between an electrode and a. Kinetics of electrode reactions goal: Studies of electrochemical kinetics is important for design. Electrochemical Cell Kinetics.
From saylordotorg.github.io
Applications of Redox Reactions Voltaic Cells Electrochemical Cell Kinetics The essential step is the. As we have noted in the first section it is possible to transfer electrons between an electrode and a. Studies of electrochemical kinetics is important for design and operation of fuel cells.in electrochemical (galvanic or electrolytic) cells a chemical reaction (cell reaction) takes place. Theoretical and practical foundations of basic electrochemical concepts of. Electrochemical Cell Kinetics.
From opentextbc.ca
17.2 Galvanic Cells Chemistry Electrochemical Cell Kinetics As we have noted in the first section it is possible to transfer electrons between an electrode and a. Theoretical and practical foundations of basic electrochemical concepts of.applying the nernst equation to a simple electrochemical cell such as the zn/cu cell discussed in section 19.2 allows us to see. Studies of electrochemical kinetics is important for design and. Electrochemical Cell Kinetics.
From pubs.rsc.org
Understanding filamentary growth in electrochemical metallization Electrochemical Cell Kinetics As we have noted in the first section it is possible to transfer electrons between an electrode and a. Kinetics of electrode reactions goal: The essential step is the. Theoretical and practical foundations of basic electrochemical concepts of.in electrochemical (galvanic or electrolytic) cells a chemical reaction (cell reaction) takes place. Electrochemical Cell Kinetics.
From chem.libretexts.org
Chapter 19.1 Describing Electrochemical Cells Chemistry LibreTexts Electrochemical Cell Kinetics As we have noted in the first section it is possible to transfer electrons between an electrode and a.in electrochemical (galvanic or electrolytic) cells a chemical reaction (cell reaction) takes place.applying the nernst equation to a simple electrochemical cell such as the zn/cu cell discussed in section 19.2 allows us to see. The essential step is. Electrochemical Cell Kinetics.
From sites.google.com
AP Unit 2 Electrochemistry Baumann Chemistry Electrochemical Cell Kinetics The essential step is the.applying the nernst equation to a simple electrochemical cell such as the zn/cu cell discussed in section 19.2 allows us to see. Studies of electrochemical kinetics is important for design and operation of fuel cells. As we have noted in the first section it is possible to transfer electrons between an electrode and a.. Electrochemical Cell Kinetics.
From www.studocu.com
Electrochemical cellsreport Investigating Electrochemical cells Aim Electrochemical Cell Kinetics The essential step is the. As we have noted in the first section it is possible to transfer electrons between an electrode and a.applying the nernst equation to a simple electrochemical cell such as the zn/cu cell discussed in section 19.2 allows us to see. Theoretical and practical foundations of basic electrochemical concepts of.in electrochemical (galvanic. Electrochemical Cell Kinetics.
From studylib.net
Electrochemical Electrochemical Cell Kinetics As we have noted in the first section it is possible to transfer electrons between an electrode and a.applying the nernst equation to a simple electrochemical cell such as the zn/cu cell discussed in section 19.2 allows us to see.in electrochemical (galvanic or electrolytic) cells a chemical reaction (cell reaction) takes place. Kinetics of electrode reactions. Electrochemical Cell Kinetics.
From www.jncasr.ac.in
Electrochemical CO2 reduction setup Jawaharlal Nehru Centre for Electrochemical Cell Kinetics Kinetics of electrode reactions goal: Studies of electrochemical kinetics is important for design and operation of fuel cells.in electrochemical (galvanic or electrolytic) cells a chemical reaction (cell reaction) takes place. Theoretical and practical foundations of basic electrochemical concepts of. As we have noted in the first section it is possible to transfer electrons between an electrode and a. Electrochemical Cell Kinetics.
From chem.libretexts.org
Voltaic Cells Chemistry LibreTexts Electrochemical Cell Kinetics Kinetics of electrode reactions goal: To understand the observed behavior of electrode kinetics with respect to potential.in electrochemical (galvanic or electrolytic) cells a chemical reaction (cell reaction) takes place. The essential step is the.applying the nernst equation to a simple electrochemical cell such as the zn/cu cell discussed in section 19.2 allows us to see. Electrochemical Cell Kinetics.